Learn the Digital nerve repair: Reconstruction with Axogen Avance processed nerve allograft surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Digital nerve repair: Reconstruction with Axogen Avance processed nerve allograft surgical procedure.
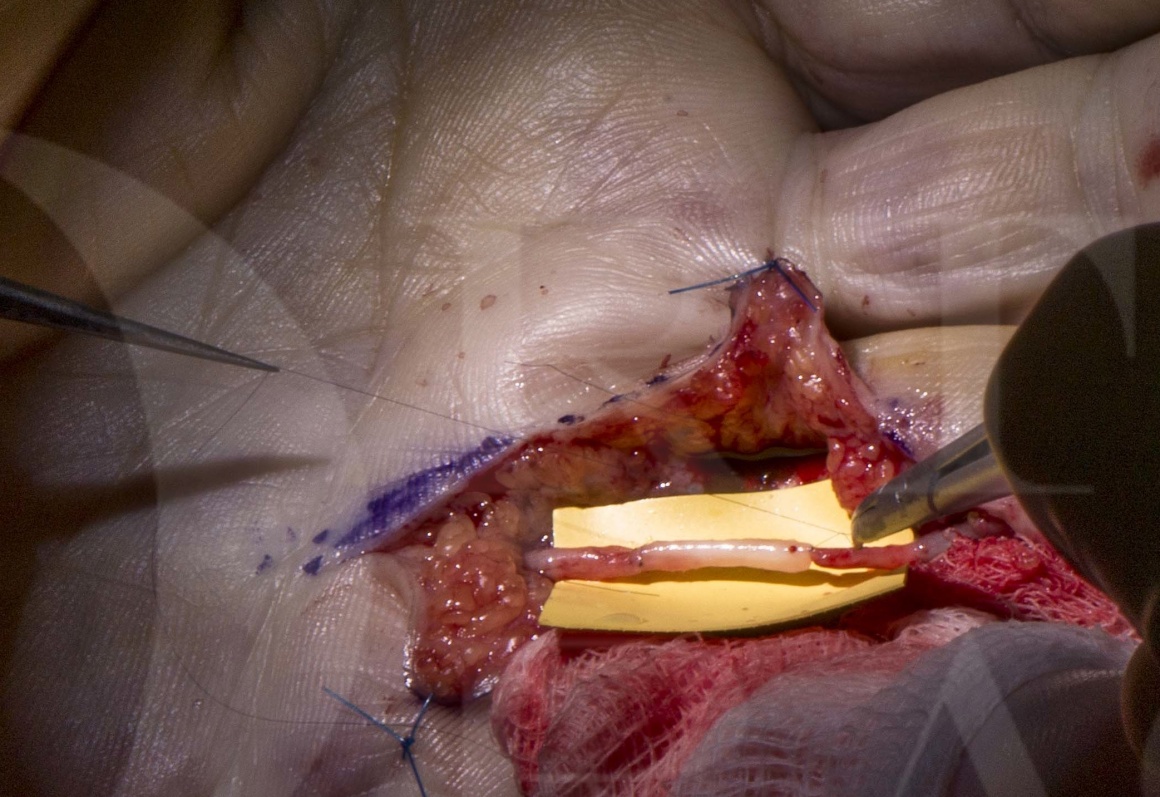
This case demonstrates the technique of bridging a small nerve gap with a processed nerve allograft.
Digital nerve injuries are a common consequence of penetrating hand injuries and are typically managed by direct microsurgical repair following exploration and wound debridement. The absence of tension across a direct epineurial nerve repair is an important factor in the success of digital nerve repair.
In many cases this is difficult to achieve due to segmental loss, scarring and retraction in missed injuries, or following the required debridement of crushed or contaminated nerve ends.
In such scenarios autologous nerve graft may be used to bridge a defect and provide an environment for nerve regeneration across a gap. The disadvantages of autograft include increased operative time and cost as well as donor site morbidity which include painful neuroma formation and sensory loss. This technique is covered elsewhere on the OrthOracle platform Nerve grafting for digital nerve defect (autograft using medial cutaneous nerve of forearm)
The search for alternative strategies to avoid autograft is ongoing. Several studies have reported upon the success of artificial nerve conduits for bridging short gaps and one such technique is covered elsewhere on the OrthOracle platform Digital nerve conduit-assisted repair with the NeurolacTM (Polyganics)
The use of nerve allograft to bridge gaps has demonstrated reasonable efficacy. Unprocessed nerve allografts will contain cellular material and immunosuppression is required until the nerve has regenerated and antigenic and cellular material has been replaced by the host. This is a major drawback and poses a significant risk of infection and other side effects from immunosuppressant therapy in otherwise healthy individuals.
Development of processed nerve allograft has removed the need for immunosuppression. AxoGen Inc is a biomedical device company that has proprietary technology for the removal of cellular debris and neuroinhibitory glycosaminoglycans (GAGs) including chondroitin from the donor nerve material. A series of enzyme washes and subequent sterilisation with gamma irradiation preserve the mechanical integrity of the endoneural tube structure of peripheral nerves. Sections of varying diameters (1-2mm/2-3mm/3-4mm/4-5mm) and lengths (15mm/30mm/50mm/70mm) are provided frozen and can be rapidly defrosted in warm saline prior to use.
The AVANCE TM processed nerve allograft (Axogen Inc. Alachua, Florida, USA) has demonstrable efficacy in short sensory nerve gaps that is comparable to autologous nerve grafts. the evidence for longer gaps and for mixed motor-sensory nerve reconstruction is more limited. A post-marketing surveillance registry study (RANGER) continues to collect results of allograft use internationally ad publishes regular reports on efficacy in the medical literature.
In the UK, the National Institute for health and care Excellence (NICE) have outlined (in IPG597) that there is enough evidence to support the use of processed nerve allograft in digital nerve gaps provided that standard arrangements are in place for clinical governance, consent and audit.
However, due to limited evidence for indications other than digital nerve repair, NICE recommend that for these cases, processed nerve allograft only be used with special arrangements for clinical governance, consent and audit or research.

INDICATIONS
A digital nerve injury must be suspected whenever a sharp laceration presents with a loss of distal sensation. The wound is then explored under magnification and the digital nerve visualised. In an acute injury, the nerve ends may be directly coapted with a tension free epineurial repair technique.
The absence of tension across a direct coaptation is an important factor in the success of digital nerve repair.
In many cases this is difficult to achieve due to segmental loss, scarring and retraction in missed injuries, or following debridement of crushed or contaminated nerve ends.
In this late presenting nerve injury, a nerve gap was bridged with processed nerve allograft.
SYMPTOMS & EXAMINATION
The commonest presentation of a digital nerve injury is following a sharp laceration to the hand. This will be accompanied by numbness at the finger tip but this may not be immediately apparent in the context of a painful laceration and preserved sensory input from other intact territories. The presence of a nerve injury should therefore be sought out by careful examination.
The absence of light touch sensation by stroking the relevant side of the pulp.
Two point discrimination is often be used. A normal sensation is reported as 1-5mm. In recent years two point discrimination has been criticised for confusing temporal and spatial stimulation and for its low sensitivty in nerve injury.
We often use the ‘ten test’ where the patient is asked to score the sensation at the pulp in comparison to an uninjured area that scores a baseline of 10/10.
In this patient where the diagnosis was made late after the wound had healed there was a clear Tinel’s sign indicating the location of the proximal nerve end.
IMAGING
Imaging is not commonly used in the diagnosis of acute digital nerve injury but in a longstanding injury where a neuroma is suspected and ultrasound scan or MRI scan may help to delineate the anatomy.
ALTERNATIVE TREATMENT
Leaving the nerve unrepaired may result in some recovery of protective sensation over 6 months but may also cause a painful neuroma at the site of injury. This may be considered in patients unfit for surgery, or unwilling to undergo wound exploration.
Other methods to bridge a nerve gap include autograft. Auto graft s typically harvested from a more expendable donor sensory nerve in the same limb. Long lengths of autologous nerve may require lateral cutaneous nerve of forearm (100-120mm), medial cutaneous nerve of forearm harvested in the upper arm (120-160mm) or sural nerve (280-350mm). Small segments (20-30mm) can use the terminal branches of the posterior interosseus nerve on the dorsal aspect of the wrist (4th extensor compartment), however there is low endometrial tube density and the donor nerve is of poor quality for critical sensory gap reconstruction in the hand. Nerve conduits may be considered for small gaps of 5-12mm although some report reasonable outcomes up to 25mm. there are numerous conduits available for short nerve gap reconstruction including collagen conduits, polycaprolactone and chitosan.
CONTRAINDICATIONS
Apart from the general contraindications to surgery, a heavily contaminated wound is not suitable for allograft until adequately debrided.

The patient underwent regional anaesthesia by means of an axillary nerve block.
A standard operating arm table and upper arm tourniquet were used.
Bipolar diathermy, micro-instruments, loupe magnification and an operating microscope are all essential.

The patient is dicharged home the same day with adequate oral analgesia and a Bradford sling to keep the limb elevated.
The digit is not splinted.
The wound is checked in an outpatient dressing clinic at 1 week and a lighter dressing is applied. Gentle finger movements are commenced at this stage to prevent stiffness.
At 10-14 days weeks the sutures are removed and the patient is allowed to return to light activities. Normal activities such as driving may be resumed between 2 and 4 weeks as pain allows.
Nerve regeneration typically takes place over a period of time that equates to 1mm of regeneration per day. In our experience regeneration through an allograft may take longer than this.
A progressive Tinel’s sign is highly suggestive of ongoing regeneration and the patient should be monitored at 6-8 weekly intervals to ensure recovery continues according to the expected timeframe. Distal sensation should be checked at each follow up appointment. Worsening neuropathic pain associated with a non-progressing Tinels suggests arrested regeneration which may merit further treatment including re-exploration.
A consistent outcome assessment of results is important. Static two point discrimination is the most commonly reported outcome measure in the literature and therefore allows comparison with other studies. As mentioned above however it is increasingly regarded as a unreliable measure. Monofilament testing may be more reproducible. A patient reported outcome measure is perhaps a more relevant measure of the success of surgery.

CHOW, S. P., & NG, C. (1993). Can a Divided Digital Nerve on One Side of the Finger be Left Unrepaired? Journal of Hand Surgery, 18(5), 629–630. doi:10.1016/0266-7681(93)90020-g
A prospective comparison of repair vs non repair of digital nerve injuries showed significantly better results in the repaired group. Improvement was demonstrated over two years post repair, with 90% achieving two point discrimination compared to only 6% in the non-repaired group.
Patients with poorer long term outcomes due to an un-repaired nerve were not subsequently willing to undergo further surgery to attempt to treat a painful digital neuroma or poor sensation.
Rinker et al. Use of Processed Nerve Allografts to Repair Nerve Injuries Greater Than 25 mm in the Hand.Ann Plast Surg. 2017 Jun;78(6S Suppl 5):S292-S295
The RANGER database is an industry registry of outcomes for processed nerve allograft. A subset analysis for digital nerve injury with gaps of 25mm or greater demonstrated recovery to S3 level in 86% of repairs which compares favourably to historical data using autograft (60-88%). The authors point out the lack of donor site morbidity or adverse reactions to the allograft.
Clare TD et al. Rehabilitation of digital nerve repair: is splinting necessary? Hand Surg Br. 2004 Dec;29(6):552-6.
This study of splinting of digital nerve repairs demonstrated no advantage to eventual sensibility. Non-splinted patients returned to work earlier and splinting was associated with increased stiffness in the early period.
Dickson et al. Nerve allograft reconstruction of digital neuromata. J Musculoskelet Surg Res 2019;3:116-22
Our own series of processed nerve allografts used to treat 12 digital neuromas in 10 patients, demonstrated satisfactory outcomes in 80%.
Taras, J. et al. Allograft Reconstruction for Digital Nerve Loss. The Journal of Hand Surgery, 38(10), 2013.
1965–1971. doi:10.1016/j.jhsa.2013.07.008
A study of allograft used to reconstruct digital nerve defects of up to 30mm demonstrating safety and clinical efficacy.
Reference
- orthoracle.com




























