Learn the Modified Oberlin double fascicular nerve transfer for elbow flexion: FCR fascicle to motor branch to biceps and FCU fascicle to motor branch to brachialis surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Modified Oberlin double fascicular nerve transfer for elbow flexion: FCR fascicle to motor branch to biceps and FCU fascicle to motor branch to brachialis surgical procedure.
Christophe Oberlin popularised the technique of nerve transfer for restoration of elbow flexion after upper brachial plexus palsy. The original technique involved highly selective fascicle transfer of a predominant flexor carpi ulnaris fascicle from the ulnar nerve to the motor branch to biceps. The technique enabled reliable and rapid re-innervation of biceps and superior results to nerve grafting for upper trunk ruptures. Modifications have included a double transfer technique using fascicles from both median and ulnar nerves transferred to the motor branches to biceps and to brachialis. I use a double technique in cases of complete elbow flexion paralysis with loss of biceps, brachialis and brachioradialis such as that resulting from a C5/6 avulsion injury or delayed presentation following rupture of the upper trunk. In cases of isolated musculocutaneous injury, proximal reconstruction for biceps can be combined with a single distal transfer to the brachialis from the ulnar nerve. The technique can also be used to salvage late presenting upper trunk or lateral cord ruptures where the time-distance phenomenon dictates that late nerve graft of a proximal rupture is unlikely to be successful or cases where proximal reconstruction has failed to provide functional elbow flexion by 9 months following injury.

INDICATIONS:
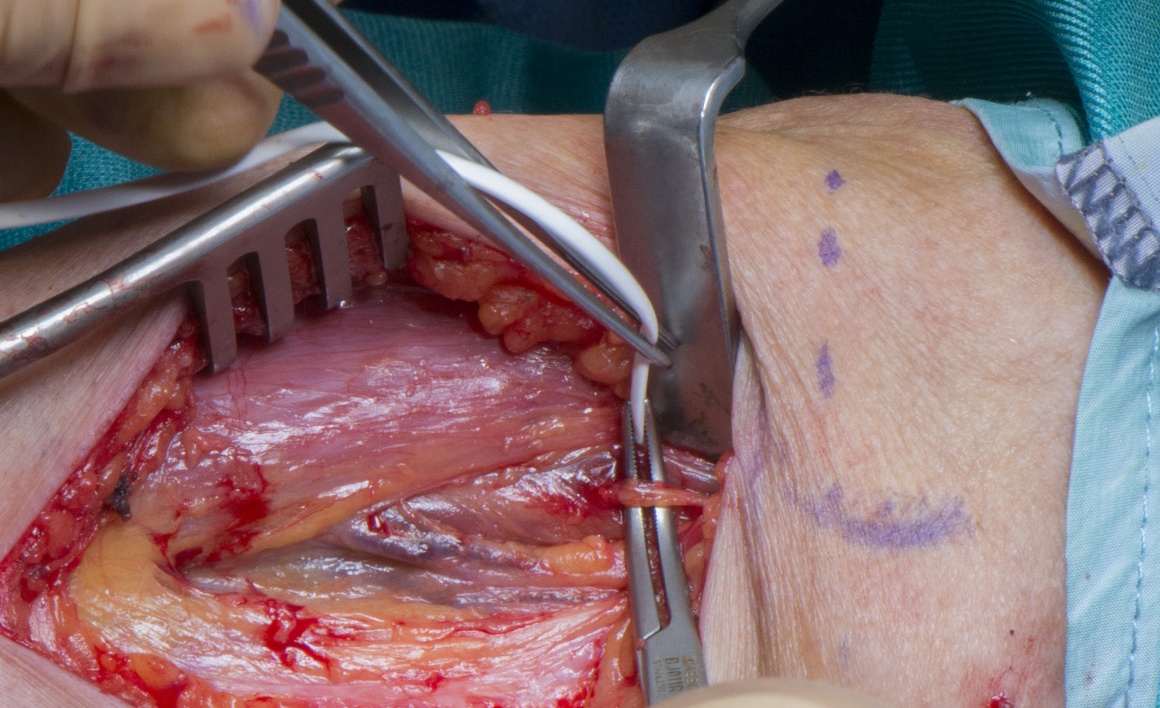
The musculocutaneous nerve (MCuN) arises from the lateral cord and contains fascicles from the anterior division of the upper trunk (C5/6). The musculocutaneous nerve supplies coracobrachialis and then passes under that muscle’s tendon 4cm inferior to the coracoid and enters the arm lying in the upper arm in the interval between the short head of biceps and coracobrachialis and at the mid-arm level in the interval between biceps and triceps. The motor branch to biceps (MBrBi) arises approximately 5cm (three finger breadths) below the anterior axillary fold and enters the medial undersurface of the muscle with a vascular pedicle. The musculocutaneous nerve continues in the interval between biceps and brachialis with a posterior and slightly lateral branch to brachialis (6 finger breadths below the anterior axillary fold) and the more anterior and medially placed lateral cutaneous nerve of the forearm. The anatomy is variable and there may be more than one branch to each muscle or a direct take off of the MBrBi may be seen from a low lying lateral cord.
The ulnar nerve (C8/T1) lies in the posterior and medial part of the interval between biceps and brachialis until the mid-arm level when it obliquely pierces the medial intermuscular septum to lie in the posterior compartment of the arm. The ulnar is difficult to identify at this level because of its intra-septal course. The first branch identified in this region is usually the medial cutaneous nerve of forearm (MCNF) from the medial cord and the ulnar nerve will be posterior to this branch. The MCNF is smaller in diameter than the ulnar nerve and has no motor action on nerve stimulation.
The median nerve (C5-T1) lies immediately adjacent to the musculocutaneous nerve at the mid-humeral level and is anterior to the brachial artery.
The Oberlin transfers are useful for reinnervation of the biceps and brachialis in C5/6 avulsion injuries when there is a complete loss of elbow flexion (ie brachioradialis is also denervated) or when recovery is poor following nerve grafting of upper trunk, lateral cord or musculocutaneous nerve ruptures or neuromata in continuity. An isolated nerve transfer to brachialis may be used when there is recovery expected to biceps but the time-distance phenomenon renders the more distally placed brachialis recovery potential limited.
The Oberlin nerve transfer to the biceps or brachialis can be successfully accomplished up to 9 months from injury. In cases of early diagnosis of avulsion of C5/6 the transfers may be completed in the first few weeks after injury as long as the donor median and ulnar nerve function is normal. When these latter nerves have sustained low grade continuity partial axonopathy injury it may be sensible to wait until the lower plexus recovery is established beyond doubt before undertaking salvage intra-plexal highly selective fascicle transfer. Reinnervation distances are short and the earliest signs of reinnervation should be seen at 2-3 months post surgery. The first sign is muscle tenderness on squeezing the recipient muscle, twitches follow with donor potentiation from 3 months with volitional control at 6-12 months. Further strengthening will occur for 2-3 years after transfer.
SYMPTOMS & ASSESSMENT:
There is loss of elbow flexion following injury to the C5 and C6 nerve roots, the upper trunk, the lateral cord or the musculocutaneous nerve. wasting of the biceps and brachialis is usually obvious, however in cases of isolated lateral cord or musculocutaneous nerve injury there may be a functional (albeit weak) preservation of elbow flexion through the brachioradialis innervated through the posterior division of the upper trunk and the posterior cord.
There is sensory loss in the cutaneous territory dictated by the level of nerve lesion. In a case of isolated musculocutaneous nerve injury the sensory loss is limited to the lateral cutaneous nerve of the forearm. In a C5 and C6 root avulsion or an upper trunk rupture there is more widespread sensory loss involving the lateral arm and volar and dorsal aspects of the radial forearm and hand. There may be dry skin and erythema due to vasodilatation due to loss of autonomic sudomotor and vasomotor function as a result of axonopathy of the affected nerves.
Careful assessment for a non-progressive Tinel at Erb’s point in the posterior triangle of the neck can confirm a non-regenerating lesion of prompt suspicion of rupture of the upper trunk. In a case of C5 ad C6 root avulsion there is no Tinel’s sign at Erb’s point due to the pre-ganglionic nature of the nerve injury. A Tinel’s sign over the infra-clavicular brachial plexus may be elicited over the lateral cord radiating to the hand or the musculocutaneous nerve radiating to the lateral forearm. A common sit eof tether and rupture of the musculocutaneous nerve is as it pierces the coracobrachialis tendon inferior to the coracoid process.
Potential donor nerves should be examined prior to consideration of nerve transfer as they may have been injured by the index accident. Thorough assessment should include MRC muscle grading of the flexor carpi ulnaris (FCU) innervated through the ulnar nerve and the flexor carpi radialis (FCR) innervated through the median nerve.
INVESTIGATION:
Imaging studies may be used to confirm a nerve root avulsion. MRI or the cervical spine and brachial plexus is useful in such cases. Ultrasound of the upper trunk can demonstrate a rupture or a neuroma-in-continuity in some cases and may be useful in obstetric brachial plexus injury cases.
Neurophysiological studies including electromyography of the biceps, brachialis and brachioradialis can confirm denervation with fibrillation. Evidence of reinnervation with polyphasia could indicate that there is some further recovery potential and an interval review may be warranted as long as the denervation is less than 9 months duration. Successful reinnervation through nerve transfers requires surgery to be completed by 9 months from injury otherwise there is irreversible loss of the intra-muscular neural plexus.
Potential donor muscles can be examined with electromyography to confirm useful function and full reinnervation if there was a concomitant involvement of the lower plexus with a continuity mixed or low grade axonal injury. This may be useful early on to plan reconstruction when there is a pan-plexus injury with a flail arm at presentation with suspicion of predominant conduction block injury of the lower plexus. However this is usually not necessary and clinical examination prior to transfer can usually confirm acceptable function and predict useful recovery from a motor nerve ransfer to the biceps or the brachialis from the median nerve, ulnar nerve or double fascicle transfer from both nerves.
OPERATIVE ALTERNATIVES:
In a rupture of the upper trunk, autologous nerve grafting can be performed to restore elbow flexion function. The distance for reinnervation is such that early surgery may restore useful biceps but the additional reinnervation distance to the nerve to brachialis means that functional recovery of the latter muscle is unpredictable. Grafting may be performed in conjunction with a nerve transfer to the more distally-placed brachialis muscle.
There are muscle-tendon transfer options in partial plexus injuries. The pectoralis major or latissimus dorsi can be transferred to restore elbow flexion although function is usually limited due to amplitude or excursion mis-match. In a patient with a weak elbow flexion from a poor continuity nerve lesion recovery the functioning common flexor muscle mass at the medial epicondyle may be transferred to the anterior aspect of the distal humerus creating a fixed flexion deformity of the elbow but allowing flexios of the elbow to be augmented by these wrist and finger flexors. This is known as a Steindler fleroplasty. Function is severely limited but it may salvage an otherwise non-functional elbow.
Late presentation of failed primary surgery can be managed with a free functioning muscle transfer using the gracilis muscle. This muscle may be anchored on the lateral clavicle and anastomosed to the thoraco-accromial vessels and reinnervated with a nerve in the vicinity, typically either a fascicle from the ulnar nerve of the medial pectoral nerve. The tendon of the gracilis can be sutured to the biceps tendon or routed to the forearm for a combined functional gain (for example elbow flexion and finger flexion; elbow flexion and wrist extension).
NON-OPERATIVE ALTERNATIVES:
There is growing interest in the use of robotic exo-skeleton devices that can be used to position the limb as an assist arm. These are most useful when there is useful retained hand function and the device improves function by allowing arm positioning in the upper limb working space. The only alternative is a flail arm orthotic device that allows locking and unlocking of the elbow joint with limited positioning options.
CONTRAINDICATIONS:
Nerve transfer should not be contemplated when there has been muscle denervation for more than 9 months or when the donor muscles are too weak to risk sacrifice of a fascicle in the ulnar or median nerves. If there is stiffness of the elbow or heterotopic bone then it is not advisable to undertake reanimation of the elbow as it will most likely fails unless there is first restoration of a full passive elbow range of motion through therapy and surgical releases.

General anaesthesia without neuromuscular blockade is required.
Use the supine position with arm table support and abduction of the shoulder to facilitate access to the medial arm. A lead hand will maintain forearm supination and elbow extension. Skin preparation should be used for the whole arm to allow visualization of the hand and wrist during nerve stimulation to ensure that no critical nerve function is at risk during fascicle selection.
A nerve stimulator, stimulator needle and electrode circuit is required to assess function in the nerves to biceps and brachialis plus for the selection of the most appropriate fascicle from the median and ulnar nerves for transfer.
Microsurgical instruments, an operating microscope and 9’0 nylon sutures are required for the dissection and nerve co-aptation.
I use Tisseel fibrin glue (Baxter) for the co-aptation to minimise sutures at the neurorraphy.

This procedure may be performed in combination with spinal accessory to suprascapular nerve transfer and nerve transfers to deltoid and teres minor from the radial nerve for C5 nerve root injuries. These procedures will be featured elsewhere in OrthOracle.
A waterproof dressing with absorbent pad is applied to the wound. The wound should be kept clean and dry for 7 days after which the patient can shower then replace the dressing. A polysling is applied to the upper limb with a torso strap around the waist before the patient wakes from surgery. The polysling and torso strap should be maintained for 3 weeks to prevent excessive passive movement at the shoulder.
During this phase the patient is encouraged to maintain isometric contraction of the donor muscles in the sling and to visualise the combination of wrist flexion and elbow flexion.
Nerve transfer rehabilitation involves a 6-phase programme of activity developed at the Centre for Nerve Injury and Paralysis, Birmingham, UK.
Phase 1 – Pre-operative phase: Education and donor optimisation. Introduction to trophic stimulation and the concept for functional electrical stimulation (FES).
Phase 2 – Protection phase: During the immediate post-operative period the nerve transfer is protected from inadvertent injury with the arm immobilised. Isometric contraction of the donor and visualisation of the combination donor-recipient action is performed during this period which typically lasts 3 weeks.
Phase 3 – Prevention phase: During this phase the arm is mobilised and neural gliding is commenced. Joint range of motion exercises (active and passive) are commenced to prevent joint contractures developing. The isometric exercises are continued and isotonic and eccentric exercises are commenced for the donor muscle to maintain function and restore strength. Functional stimulation can be commenced on the donor muscle. Trophic stimulation can be maintained on the recipient muscle.
Phase 4 – Power phase: During this period the donor muscle is strengthened and the recipient muscle starts to respond. Typically the first sign of reinnervation is a tender muscle squeeze sign due to small fibre reinnervation. Typically this is 3 months following transfer but is affected by the distance of the co-aptation from the recipient motor point.Visible flickers of contraction follow within 6 weeks and donor activation potentiate the recipient response. FES continues and the phase lasts for approximately 6-12 months during which useful motor grade returns: Medical Research Council – (MRC) Grade 3-4.
Phase 5 – Plasticity: During this phase the patient works on activation of the recipient muscle without activation of the donor. This phase can overlap with phase 4 and is guided by a therapist specialised in nerve transfer rehabilitation.
Phase 6 – Purpose: During this phase the patient introduces function tasks discussed as objectives during the pre-operative phase. This period of training is tailored to the individual and includes work hardening. Improvements are typically found in power and functional performance for at least 2 years following nerve transfer surgery.
Outcomes assessment can be with MRC scales but because of the huge functional variation within MRC Grade 4, I prefer absolute muscle testing with digital myometry and comparison with the contralateral limb. In addition fatigue testing and functional scores are important. The EQ5D, DASH and the BrAT scores are in common use in assessing upper limb function after brachial plexus injury. The Canadian Occupational Performance Measure (COPM) is an ideally suited tool to assess patient specific objectives and outcomes.

History of the Oberlin nerve transfer procedure:
Adolf Stoffel described the technique of triceps to musculocutaneous nerve transfer via an anterior approach in 1911 in a textbook of orthopaedic surgery “Orthopadische Operationslhere” published in 1913. This text was never translated from German and the technique did not receive wide uptake.
Christophe Oberlin described the technique of ulnar nerve fascicle transfer in 1994 with a follow-up series in 1997. In 2002 Oberlin wrote a technical description of the procedure and it gained in popularity.In 2005 Susan Mackinnon published a technique of double nerve transfer for elbow flexion and this was followed by a paper from Oberlin in 2006 detailing the same technique. Bertelli in 2010 wrote about the role of adjunctive distal nerve transfer after proximal nerve repair or graft. The current practice is for a single FCU fascicle to biceps or in a double transfer to use the FCU fascicle for brachialis and the FCR fascicle to biceps in my practice. In caes of anticipated biceps recovery from proximal surgery I undertake a distal concomitant nerve transfer FCU to brachialis in keeping with the rational described by Jayme Bertelli.
Author’s note:
Double nerve transfer at the shoulder for abduction (deltoid and supraspinatus) and for external rotation (infraspinatus and teres minor) confers superior results in terms of brachiothoracic motion and strength with MRC grade 4 shoulder abduction in approximately 80% of patients compared to around 80% MRC grade 3+ with a single transfer for each function. The benefit of the second transfer at the elbow is less clear with approximately 70% achieving MRC grade 4 after a single transfer and 90% MRC grade 4 after a double transfer. This is based on the author’s own series of nerve transfers around the shoulder and elbow and is supported by the published literature. The single transfer should use the UN as fascicle donor. The added benefit of 20% increase MRC grade 4 should be weighed against the risk of sensory compromise to the hand and reduction in wrist flexion strength when the median nerve is used as a second donor.
Avoid dissection in the donor nerve by surface mapping of the topography using the nerve stimulator and exposing only the target area. Do not undertake intra-fscicle dissection until a donor is identified and consider minimizing the length of the intra-fascicle dissection by using FCU fascicle to brachialis and FCR fascicle to biceps when a double transfer is selected (MacKinnon modification).
Use caution when there is C7 involvement as the FCR will not be of sufficient strength for a reliable double transfer technique and I would recommend a single FCU fascicle to the MBrBi as the preferred choice in such cases. The rationale here is that as the injured is more extensive there are fewer donors available and more potential targets and so a pragmatic approach to reconstruction is required and it should be accepted that the outcome is likely to be inferior to that following isolated C5/6 avulsion injury.
References:
Orthopadische Operationslhere. “Operationen am nervensystem” Oskar Vulpius and Adolf Stoffel. Publshed by Ferdinand Enke, Stuttgart, 1913
Nerve transfer to biceps muscle using a part of ulnar nerve for C5-C6 avulsion of the brachial plexus: anatomical study and report of four cases.
Oberlin C, Béal D, Leechavengvongs S, Salon A, Dauge MC, Sarcy JJ.
J Hand Surg Am. 1994 Mar;19(2):232-7.
Ulnar nerve fascicle transfer onto to the biceps muscle nerve in C5-C6 or C5-C6-C7 avulsions of the brachial plexus. Eighteen cases].
Loy S, Bhatia A, Asfazadourian H, Oberlin C.
Ann Chir Main Memb Super. 1997;16(4):275-84. French.
Restoration of elbow flexion in brachial plexus injury by transfer of ulnar nerve fascicles to the nerve to the biceps muscle.
Oberlin C, Ameur NE, Teboul F, Beaulieu JY, Vacher C.
Tech Hand Up Extrem Surg. 2002 Jun;6(2):86-90
Preliminary results of double nerve transfer to restore elbow flexion in upper type brachial plexus palsies.
Liverneaux PA, Diaz LC, Beaulieu JY, Durand S, Oberlin C.
Plast Reconstr Surg. 2006 Mar;117(3):915-9.
Results of reinnervation of the biceps and brachialis muscles with a double fascicular transfer for elbow flexion.
Mackinnon SE, Novak CB, Myckatyn TM, Tung TH.
J Hand Surg Am. 2005 Sep;30(5):978-85.
Double fascicular nerve transfer to the biceps and brachialis muscles after brachial plexus injury: clinical outcomes in a series of 29 cases.
Ray WZ, Pet MA, Yee A, Mackinnon SE.
J Neurosurg. 2011 Jun;114(6):1520-8. doi: 10.3171/2011.1.JNS10810.
Nerve root grafting and distal nerve transfers for C5-C6 brachial plexus injuries.
Bertelli JA, Ghizoni MF.
Reference
- orthoracle.com














































































